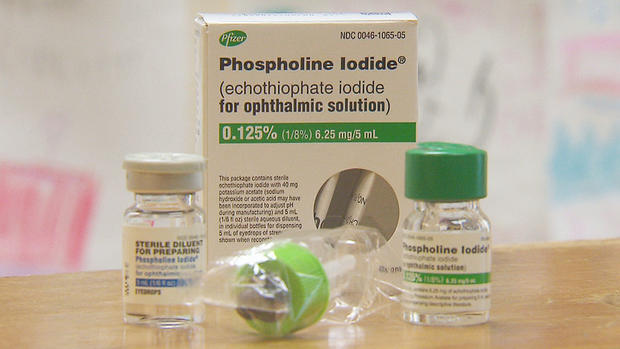I-Team: Parents Relieved 'Miracle' Eye Drops Will Still Be Available For Son With Glaucoma
BOSTON (CBS) - Joe and Holly Decker are overwhelmingly relieved that Pfizer's sight saving eye drops their four-year-old son Tate needs will now still be available. Holly Decker says she cried when she got the news. Joe, Tate's dad tells the I-Team his prayers were answered. "It was great to see Pfizer step up and follow through," he said.
The I-Team reported in May that Pfizer was taking the phospholine iodide drops off the market- leaving families like the Deckers with the possibility that their children could face blindness and risky surgery. It's something Holly says has been scary.
But on Monday, Pfizer reversed course. In a statement Pfizer said it got FDA approval to extend the shelf life of the inventory it has of the phospholine iodide drops to 12 months and is working with another drug company to help manufacture the medication that Holly calls, "our miracle drug."
Tate suffers from childhood glaucoma. The disease has no cure but can be controlled with Pfizer's daily drops that he and hundreds of others use to keep their sight.
He is reading and knows his letters and colors- something his family says wouldn't have been possible without the ability to see.
Full statement from Pfizer:
We have been working with the utmost urgency exploring several options to return P.I. to the market for patients, and consequently, we have a path forward to improve short and long-term availability.
In the short-term, the U.S. Food and Drug Administration (FDA) approved our request for a 12-month extension of the shelf life of previously manufactured P.I. that is currently available in our inventory. We believe this will provide approximately six months of supply in the immediate future.
To address the long-term supply, we have completed an agreement to immediately transfer P.I. to Fera Pharmaceuticals, which has the capabilities, experience, and motivation to bring P.I. to patients on a sustainable and expedited basis. Led by a seasoned executive team including Frank DellaFera, former CEO of Sandoz US, Fera was selected out of a pool of candidates based on multiple factors, including expertise and focus in ophthalmic medicines, experience in bringing complex medicines to patients, and their understanding of the regulatory process for ophthalmic medicines.
As part of the agreement, Pfizer will transfer the New Drug Application for P.I., the P.I. trademark, and existing inventory to Fera Pharmaceuticals. Additionally, Pfizer will provide technical consultation services to Fera to help progress the development and manufacturing of P.I. in an expedited manner. Fera is prepared to complete the transition as soon as possible and has experience working with the FDA to expedite product approval and subsequent return to market for products that are complex to manufacture.
We are optimistic that Fera will be able to progress the development and manufacturing of P.I. in an expedited manner with a reliable and compliant manufacturing supply chain.