Weymouth Woman Fears Another Bout With Cancer After Tissue Expanders Recalled
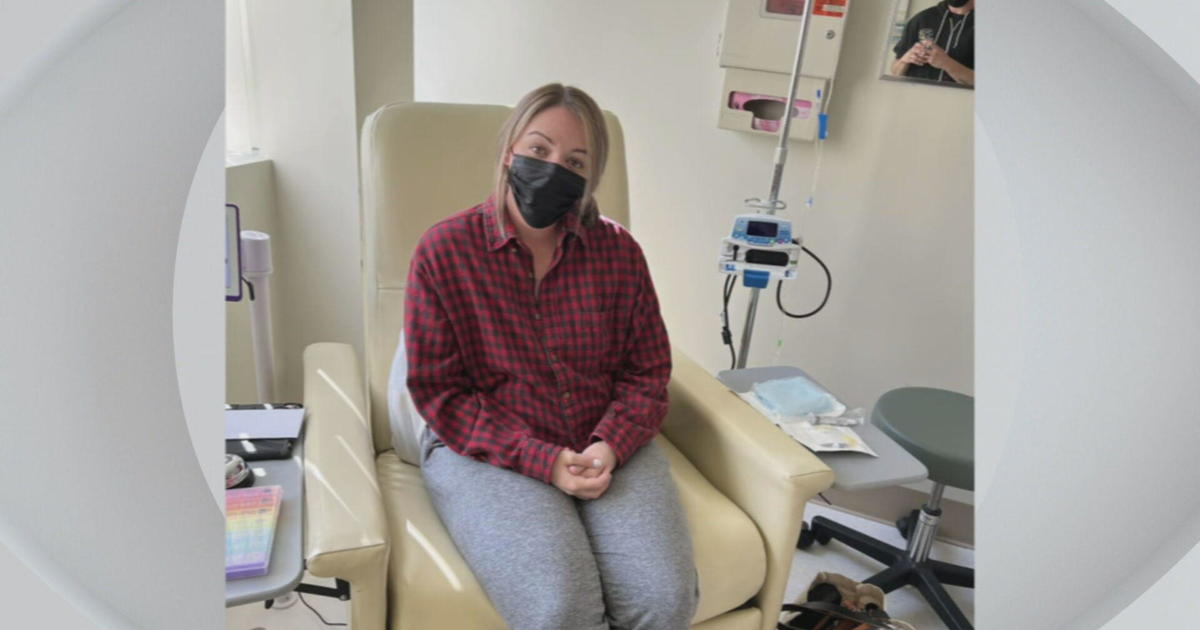
WEYMOUTH (CBS) - Kelly Cassier's life was starting to get back to normal after the 43-year-old mother of two was diagnosed with stage 3 breast cancer two years ago.
Until yesterday, when she found out something in her body could be killing her.
"I'm sad because my kids are starting to enjoy their mom again. They certainly weren't enjoying me for a long time. I wasn't enjoying me. Now, I don't want to start over," Cassier said.
Cassier still takes a chemotherapy pill daily and had a double mastectomy shortly after her diagnosis. That's when her doctors placed tissue expanders in her chest.
"They put them in when you have your double mastectomy to push the skin for your final reconstruction surgery," she explained.
This week the FDA issued a recall on specific types of textured breast implants made by the company Allergan. The implants have been linked to a rare form of lymphoma.
The FDA says there have been 573 cases as of July 6, 33 of them leading to death.
In that same recall were certain tissue expanders, like the ones inside Cassier.
"I am angry any woman has to worry about this right now," she said.
In the recall, the FDA wrote, "The FDA does not recommend removal for patients without symptoms due to potential risks...." The most common symptoms are "persistent swelling or pain in the area of the breast implant."
Cassier's breast reconstruction surgery is scheduled for mid-September at Brigham and Women's Hospital. That's when she will also have those potentially dangerous tissue expanders removed. This surgery was supposed to mean her battle with cancer was close to an end. While she waits, she's left to wonder if her fight will start all over again.
"Is today day one of my second bout with cancer? I don't know. I don't know."
Click here for more information on the recall.