FDA Approves Clinical Trial Of Trauma Foam In Patients At MGH
BOSTON (CBS) - The FDA has approved the use of a potentially lifesaving foam on patients in a clinical trial.
"It's remarkable and it's very exciting for us," said Dr. David King, a trauma surgeon at Massachusetts General Hospital.
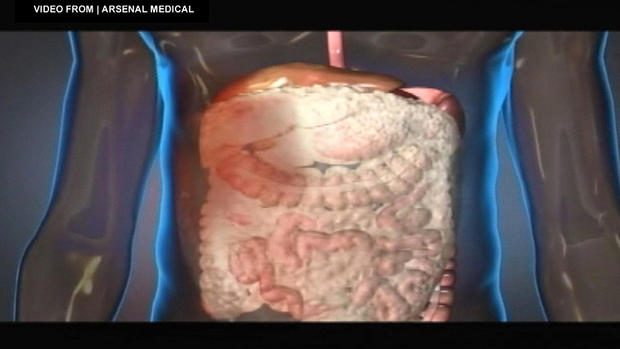
Called ResQFoam, it was developed by Dr. King and a team from Arsenal Medical in Watertown, to quickly stop internal bleeding.
Now, the FDA says it can be used on up to 40 patients at Mass. General.
"To allow us to intervene and stop, temporarily stop bleeding patients essentially the moment they arrive in the emergency department," King said.
The foam is injected into the body through the belly button and expands in the abdomen.
"It wraps around injured tissues and injured organs and puts gentle pressure on them which temporarily slows or stops hemorrhage," King said.
An Army medic, King knows how critical time is when it comes to blood loss. He has treated fellow soldiers on the battlefield and victims of trauma on the home front.
"When you're suffering from internal bleeding, we don't know how much time you have because no one can see what types of injuries you have on the inside."
King hopes ResQFoam will give many more patients a fighting chance.
Anyone who wants to opt out of the clinical trial at MGH can obtain a special bracelet. More information can be found at resqfoam.com.